St Anne’s Academic Review 8 – 2018
Understanding the Paleofluid Records: a Case Study of Southern Utah
Rebecca Tyne, Department of Earth Sciences
STAAR 8 – October 2018, pp. 87-96
——————————–
Published: 03 Oct 2018
Review process: Open Peer Review
Draft First Uploaded: 26 Jul 2018. See draft and reviewers’ comments.
Abstract
The ability to understand subsurface fluid-rock systems is critical to the development of geological carbon dioxide sequestration and hydraulic fracturing. The Colorado Plateau in the United States of America is an archetypal location for diverse fluid flow. Across the plateau, especially in southern Utah, the Jurassic Navajo sandstone has remarkable red and white colour variations, which are a result of sandstone bleaching. This spectacularly preserved fluid flow event, accompanied with extensively drilled wells and the well characterised geological history, means that southern Utah is an ideal natural laboratory for studying the paleoflow. The type of the fluid flow responsible for the bleaching is highly debated; the main candidates are a huge exhumed hydrocarbon field bigger than those in Saudi Arabia, a natural carbon dioxide flow or hydrogen sulfide. The spatial and temporal interaction of simple, independent factors, as seen in the Colorado Plateau, leads to complex results not relatable to any individual process. This article critically reviews the evidence for paleo-hydrocarbon, carbon dioxide and hydrogen sulfide flows across the basin. It also suggests that future paleofluid flow studies, both in this region or across the world, need a whole basin-scale perspective in order to truly understand the interaction between the fluid flow and the subsurface.
Introduction
Recent debates over hydraulic fracturing and its link to seismicity and geological carbon dioxide sequestration have resulted in an urgent need to better understand subsurface fluid and paleofluid flow and interactions. This would allow the development of natural analogues for these modern systems. In addition, there is an increasing awareness about the role of subsurface fluids in connecting the lithosphere with the critical zone (the near-surface environment where the interactions of rocks, fluids, atmosphere and biological organisms control and regulate the availability of resources needed for life to exist). As a result, an understanding of subsurface fluid-rock systems is becoming increasingly important. The subsurface migration for specific types of fluids over short timescales has been extensively studied in order to assess and manage groundwater, hydrocarbon and ore deposit resources. However, fewer studies have explored multiple fluids within a flow, and how these fluids interact. An understanding of these interactions during flow, especially in areas of rock deformation, could aid in the human management of subsurface resources. Consequently, a suitable natural laboratory is required to explore and understand the connection between paleofluid flows and the lithosphere and critical zone.
The Colorado Plateau in the United States has some of the most iconic and controversial records of diverse fluid flow in the world, which have been recorded in the rock record as extensive bleaching of the upper sandstones and remarkable colour variations (see Figure 1). This makes the Colorado Plateau an ideal natural laboratory for studying paleofluid flow. The incised 3D exposure in southern Utah, extensively drilled wells, subsurface samples, and a relatively well-characterised geological history make it especially well suited for studying paleofluid flow. The spatial and temporal interaction of simple, independent factors seen in the Colorado Plateau leads to complex results which are not relatable to individual process. Therefore, this article will look at fluid responsible for this bleaching in this basin as well as the need for a basin-system-scale perspective to truly understand how both the reservoir and rocks have responded to paleofluid flows.

Figure 1: Exposure of the bleached Jurassic sandstones in southeastern Utah, near Moab. Cross stratification from dunes (CB) can be seen in both bleached (B) and unbleached units (R).
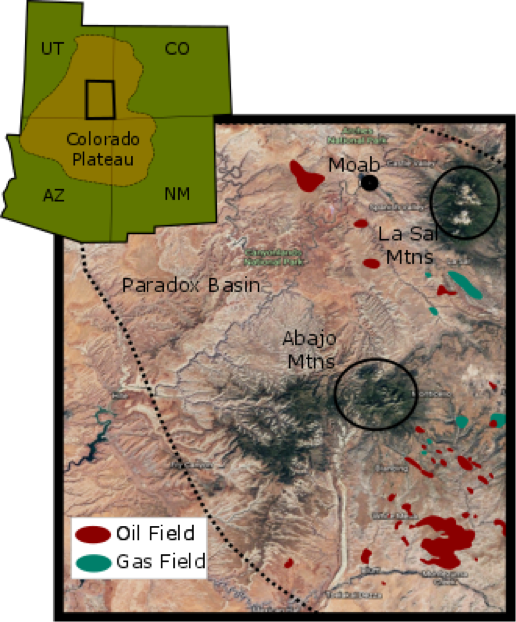
Figure 2: A map of the broader Four Corners Region of the U.S.A and a more detailed map of the study area. Satellite image is taken and modified from Google Earth; Oil and Gas Fields are adapted from assets.geoexpro.com.
Geological Setting
The Colorado Plateau is located in the four corners region (where the states of Arizona, Utah, Colorado and New Mexico meet) in the southwest of the United States (Figure 2). It covers an area of approximately 50,000 km2. The plateau’s interior is largely unaffected by significant tectonic deformation. Southern Utah is an archetypal location for paleofluid flow, made up of a thick Palaeozoic-Mesozoic sedimentary sequence. The Jurassic to Cretaceous system rocks make up the main sedimentary sequence. They consist of thick marine and non-marine sequences formed from erosion, especially resulting from during the Nevadian and Sevier orogenies to the west of the Colorado Plateau. The Jurassic units are generally flat lying and comprise of four main aeolian units affected by diagenetic iron. In ascending order, these are the Navajo Sandstone, Page Formation, Entrada Formation and the Summerville/Morrisson Formation [1, 2, 3, 4, 5]. The Navajo sandstone and its equivalent units are dominated by large scale high angle aeolian cross stratification (Figure 1) [6]. Together, these units form the largest dune field preserved in Earth’s history [1]. The Navajo Sandstone is a well sorted, fine-medium grained quartz arenite that was oxidised during diagenesis [6] and it is the main aquifer unit in some areas of the plateau. The well-preserved porosity and permeability of the Navajo sandstone most likely allowed for large fluid flows and the resultant bleaching through the unit [7]. The Page formation is local to the Moab area and it is only a few metres thick. It is a basal chert–pebble conglomerate and fines upwards to a coarse-grained sandstone [4]; this unit also has a high permeability. The Entrada formation contains three different members (Dewey Bridge, Slick Rock and Moab Tongue) with differing lithologies and characteristics affecting the fluid movement. The Dewey Bridge Member is an interbedded sandstone, siltstone and mudstone with local bed scale breccia. The Slick Rock Member is a largely aeolian sandstone, sea-dune deposit with a moderate permeability [3, 8]. The Moab Tongue member is local to southern Utah and pinches out to the south and west of the Moab. It is a relatively thin fine-grained unit which is commonly jointed and contains cross-stratified aeolian dune sets. The permeability is similar to that Navajo Sandstone. The Summerville formation and Tidwell member of the Morrison formation are both very thin-bedded sandstones and mudstones overlying the Moab Tongue. They are non-calcareous red beds from a marine incursion of sandstone in a coastal to tidal setting separated by an unconformity; these represent the confining layer in the system and have remained red [4].
The Colorado Plateau includes salt tectonics, monoclinal folds and broad flexures, faults, and igneous laccoliths and volcanic features. The Laramide orogeny which occurred during the early Tertiary resulted in uplift of the Colorado Plateau, monoclinal folding and minor faulting, which link to regional faults at depth [9]. The thick salts of the Paradox sub-basin have added additional structure to the Colorado Plateau by deforming and folding the overlying structures and creating eight salt anticlines, the crests of which were eroded prior to burial. The salt diapirs have subsequently collapsed, creating valleys at the centre of the anticlines. Between 6 million and 1200 years ago, the edge of the plateau was subject to volcanic activity which created volcanic extrusive features, laccoliths as well as natural CO2 fields.
Bleached Sandstone
The Jurassic sandstones of southern Utah were stained red during early diagenesis, as a result of the release of iron from detrital minerals and subsequently oxidized to form hematite grain coatings or iron cements [10, 11]. Today the rocks that remain red represent the least altered parts of the formations. However, large areas of the region have been bleached as a result of a reducing paleofluid flow [6]. The Navajo and Entrada sandstones are both heavily affected by this bleaching [4, 10, 12]. There is also minor bleaching of the Permian White Rim sandstone [13] and the Triassic Moenkopi Formation [14]. The bleached sandstone tends to be at the top of formations suggesting that the responsible fluid is buoyant. The bleaching also cuts across stratigraphic and petrographic boundaries. The contacts between the bleached and unbleached zones are sharp which could suggest significant burial of rocks before bleaching [15]. This is consistent with rapid subsidence of up to 10 km during the Cretaceous [16].
Although bleaching occurs across the entire plateau, the most continuous and extensive bleaching is in southern Utah at the crest of steeply dipping Laramide uplifts and monoclines. There is also a spatial relationship between the iron oxide deposits and faults in the region (especially the Moab fault) [4] indicating that faults in the region were conduits for the reducing fluids that bleached the sandstones.
Iron oxide deposits appear as concretions in the bleached zones and throughout the Jurassic stratum. These concretions vary in shape and size from millimetres to centimetres in scale and cut across bedding planes. The iron is also deposited as hematite columns and pipes (tens of centimetres in diameter and several meters long) and erosionally resistant towers (up to tens of meters high e.g. at Duma Point). The red staining of the hematite columns decreases toward their cores and they can have both sharp and diffuse edges. The type of edge is dependent on the groundwater flow: in the places where water streaks the hematite column the edges become diffuse and resemble ‘Comet Tails’, which could indicate paleo flow direction [4].
Detailed geochemical studies of the bleached zones show they are depleted in 18O and 13C suggesting that a reducing fluid ascended through faults and mixed progressively with younger groundwaters. The fluid subsequently became oxidising, causing deposition of calcite, copper and other minerals (see Figure 3) [4, 11, 17, 18]. There is evidence that a high salinity brine ascended through the faults from either the Upper Palaeozoic aquifer or that it resulted from evaporate dissolution [19]. This brine may have been associated with the reducing agent or may have ascended during a separate event; and at least two different fluids have been found to have ascended up the Moab Fault [4].

Figure 3:
Hand specimen showing copper mineralisation in linear planes as a result of fluid flow along boundaries.
The fluid flow responsible for this bleaching is highly debated. The main candidates are a huge exhumed hydrocarbon field or a natural carbon dioxide flow. Both of these options will be discussed below.
Possible bleaching fluids
In order for the sandstone to be bleached, and to explain the precipitation of uranite, pyrite and pyrite pseudomorphs, the iron grain coatings must have been reduced and mobilised by a fluid [18]. As the fluid migrated, it must have been both stratigraphically and structurally controlled. Possible iron reducing fluids that could be responsible for the colour change are: hydrogen sulphide (H2S); hydrocarbon; CO2; methane; and organic acids [4, 20]. This mobilised iron can be seen to have travelled up to several kilometres prior to precipitation.
Similar bleaching is observed in Montana and is a result of the direct contact of hydrocarbons with iron. It has been suggested that this could also be the cause of the bleaching in southern Utah [20, 21, 22, 23, 24]. Experiments by Chan et al. confirmed the ability of hydrocarbons to bleach sandstones [4]. They found that hydrocarbons, in the presence of an acid, reduce and mobilise the iron, producing CO2 and water as by-products (Eq. 1). Organic acids and methane which may also be present in hydrocarbons can also reduce the iron and release CO2 and water (Eqs. 2 and 3 respectively). The chemical equations that govern these processes (taken from Chan et al. [4].) are given below.
CH2O + 2Fe2O3 + 8H+ = CO2 + 5H2O + 4Fe2+ (Eq. 1)
CH3COOH + 4Fe2O3 +16H+ = 2CO2 + 8Fe2+ + 10H2O (Eq. 2)
CH4 + 4Fe2O3 +16H+ = 10H2O + CO2 + 8Fe2+ (Eq. 3)
Based on bleached rock volumes and on pore volumes, it is estimated that 18.5×1012 barrels of oil would have been needed prior to the erosion to cause the observed bleaching. This figure would have made it the world’s largest hydrocarbon field (currently, the largest field is the Ghawar field in Saudi Arabia which contains approximately 1×1010 barrels) [25]. This is not an unfeasible estimate for southern Utah as several hydrocarbon fields in the plateau would have reached maturity during the late Cretaceous [14, 26, 27]. The timing of these fields maturing coincides approximately with the Laramide orogeny and the beginning of the sandstone bleaching. The anticlines associated with the location of bleaching would have also provided major structural traps for this paleo hydrocarbon reservoir. Furthermore, bitumen veins and tar sands within the bleached sandstone provides additional evidence that hydrocarbons were once present. Bitumen veins occur across the region with the highest concentration within 250 m of the Moab fault. They have bleached the edges of the sandstone they are in contact with, confirming their ability to cause the bleaching seen [6]. Tar sands occur in the Slick Rock Member of the Entrada formation and are up to 9 m thick [4]. The depleted 13C signature seen in the calcite veins and cement of the sandstones can be attributed to carbon exchange with hydrocarbons which can cause decarboxylation [4, 6]. This again confirms the viability of hydrocarbons as the reducing fluid and paleo-fluid flow responsible for the bleaching of the sandstones.
However, bitumen is not found in all bleached layers and there is currently no indication as to what happened to the remaining hydrocarbons [20]. Additionally, to explain the preferential bleaching at the top of formations, hydrocarbons would have had to migrated through the formations as a buoyant fluid and therefore, they would not have been constrained to down dip directions. However, the comet tails on the hematite pipes suggest a single down dip flow direction [20]. The flow directions suggested by the comet tails are inconsistent with hydrocarbons as the responsible reducing agent. Chan et al. suggested that multiple fluids —one reducing fluid and one that later caused the oxidation— might have migrated up the faults [4]. Nevertheless, the single flow direction seen from comet tails make it unlikely that these fluids would have occurred as distinct episodes with multiple flow directions. This again suggests that it is unlikely that hydrocarbons alone could be responsible for the extensive bleaching.
Loope et al. proposed that the reducing nature of the fluid could have been instead a result of dissolved CO2 [20]. Within the pre-Triassic strata in the Colorado Plateau there are eleven CO2 fields [28, 29, 30], and CO2 springs are associated with local faults [31]. The abundance of CO2 in the region indicates that CO2 could be responsible for the reducing groundwater. Carbon dioxide could have seeped through the Triassic sealing sediments via faults and interacted with the groundwaters [29]. The depleted 13C ratio observed by Chan et al. could also be explained by upwelling of dissolved CO2 [4]. However, laboratory experiments indicate that CO2 does not cause the bleaching of the iron in sandstones. Though, CO2 was able to aid in the mobilisation of large amounts of iron from fractured minerals suggesting that it is a possible source of iron in modern pore fluids [31]. Therefore, the presence of a reducing agent is required alongside CO2 to dissolve the hematite [31, 32].
Alternatively, hydrogen sulphide on its own (as opposed to mixed with hydrocarbons) could be the cause of the bleaching. It is abundant in southern Utah as evidenced by cold H2S seeps (especially within Salt Valley), pyrite mineralisation and very H2S rich brines in the formation underlying the Dolores River. Hydrogen sulphide could then be brought up from the brines to the sandstones via faults. Purser et al. have conducted preliminary experiments to determine the viability of H2S as the sole cause of iron bleaching, which could be applied to the area (Eq. 4) [31].
2H2S + Fe2O3 + 2H+ = FeS2 + 3H2O + Fe2+ (Eq. 4)
Hydrogen sulphide could be sourced from the interaction of groundwater with gypsum, within the paradox salt formation, or from the reaction of thioacetamide and water [41]. Preliminary results show H2S to have five times the reducing power of dissolved hydrocarbons [31]. However, the volumes of H2S across southern Utah and the Colorado Plateau, are unlikely to be large enough to cause the amount of bleaching seen, especially as the salt is localised to Paradox Basin and not widespread. Therefore, it is likely that H2S could have caused some of the sandstone bleaching, especially in southern Utah, but other agents must also have been present to cause the extent of bleaching seen.
Conclusion and additional work required
In conclusion, it seems unlikely that any of the fluids so far proposed (hydrocarbons, CO2 and H2S) are solely responsible for the bleaching of the sandstone in southern Utah. This is mainly due to the volumes of fluid that would be required at a given reducing power. Hydrocarbons seem to be the most plausible cause for the bleaching because it seems feasible that they could reach the required volume. However, whilst hydrocarbons appear to play a key role, they are not the only contributing factor and it is likely that the bleaching was caused by a combination of hydrocarbons, CO2, H2S and other agents.
In southern Utah the first step to understanding the complex paleoflow in the area to is to determine the fluid responsible for the bleaching. However, to understand the full paleofluid flow history of the area and in other regions of the world, the sedimentation, deformation and lithological change through history also need to be well characterised. This will allow for the development of conceptual models to understand system-scale fluid evolution and the formation and management of resources. An understanding of the modern fluid flow in the basin including sources, residence times, and flow paths, will provide additional insight into the basin characteristic and possible constraints on the paleoflow. This could be determined by the use of several techniques including noble gases, radiocarbon, stable and clumped isotopes and strontium isotopes. This broader outlook will go beyond determining specific fluid types and restricted spatial and temporal perspectives to elucidate long-term and possible evolutionary fluid flow relationships. Insights from this will improve the understanding critical zone interactions, and aid in resource management and the further development of carbon capture and storage.
References
[1] R.C. Blakey, F. Petrson, G. Kocurek., “Synthesis of late Palaeozoic-Mesozoic aeolian deposits of the Western Interior of the United States” Sedimentary Geology, Vol. 56, pp. 3-125, 1988. PARRY 2007
[2] R.C. Blakey “Paleogeographic and tectonic controls on some lower and middle Jurassic erg deposits, Colorado Plateau” in Mesozoic systems of the Rocky Mountains region, M.V. Caputo, J.A. Peterson and K.J. Franczyk, Eds. Denver: Rocky Mountain Society for Sedimentary Geology, 1994, pp.273-298.
[3] W.T. Parry, M.A. Chan, B. Beitler., “Chemical Bleaching indicates episodes of fluid flow in deformation bands in sandstone” AAPG Bulletin, Vol. 88, pp. 1-17, 2004.
[4] M.A. Chan, W.T. Parry, J.R. Bowman., “Diagenetic Hematite and Manganese Oxides and Fault-Related Fluid Flow in Jurassic Sandstones, south eastern Utah.” AAPG Bulletin, vol. 84, No. 8, pp. 1281-1310, Sep 2000.
[5] W.T. Parry, C.B. Forster, J.P. Evans, B.B. Bowen, M.A. Chan., “Geochemistry of CO2 sequestration in the Jurassic Navajo Sandstone, Colorado Plateau, Utah” Environmental Geosciences, Vol. 14, No. 2, pp. 91-109, June 2007.
[6] B. Beitler, M.A. Chan, W.T. Parry., “Bleaching of Jurassic Navajo Sandstone on Colorado Plateau Laramide highs: Evidence of exhumed hydrocarbon supergiants?” Geology, Vol. 31, no. 12, pp. 1041-1044, Dec 2003.
[7] L.E. Sprangler, D.L. Naftz, Z.E. Peterman., “Hydrology chemical quality and characterization of salinity in the Navajo aquifer in and near the Greater Aneth oil field, San Juan County, Utah” USGS Water Resources Investigations Report, pp.90, 1996.
[8] M.A. Antonellini, A. Aydin., ” Effect of faulting on fluid flow in porous sandstones: Geometry and Spatial distribution” AAPG Bulletin, Vol. 79, pp. 642-671.
[9] G.H. Davis, “Structural Geology of the Colorado Plateau region of Southern Utah with special emphasis on deformation bands” Geological Society of America Special Paper, Vol. 342, pp=157, 1999.
[10] T.R. Walker, “Red beds in the western interior of the United States” USGS professional paper 853, pp. 49-56, 1975.
[11] T.R. Walker, E.E. Larson, R.P. Hoblitt., “Nature and origin of hematite in the Moenkopi Formation (Triassic), Colorado Plateau: A contribution to the origin of magnetism in red beds” Journal of Geophysical Research, Vol. 86, No. B1, Jan 1981.
[12] I.R. Garden, S.C. Guscott, S.D. Burley, K.A. Foxford. J.J. Walsh, J. Marshal., “An exhumed paleo-hydrocarbon migration fairway in a faulted carrier system, Entrada Sandstone of SE Utah.” Geofluids, Vol. 1, pp. 195-213, 2001.
[13] R. Sanford, “Ground water flow and migration of hydrocarbons to the lower Permian white rim sandstone tar sands triangle, south eastern Utah” USGS Bulletin 2000-J, pp. 24. 1995.
[14] R.C. Blakey, “Stratigraphic and depositional analysis of the Moenkopi Formation South-Eastern Utah” USGS Bulletin 104, pp. 53-54, 1974.
[15] H. Chu., “Geochemical and paleo-geothermal studies of the basinal fluids in the Athabasca Basin- Implications for unconformity-related uranium mineralization” Ph.D. dissertation, Dept. Geology., Univ. of Regina., Regina, Canada, 2016.
[16] B. Beitler, W.T. Parry, M.A. Chan., “Fingerprints of Fluid Flow: chemical diagenetic history of the Jurassic Navajo Sandstone southern Utah, USA” Journal of Sedimentary Research, Vol. 75, pp. 547-561, 2005.
[17] S.J. Morrison, W.T. Parry., “Formation of carbonate-sulfate veins associated with copper ore deposits from saline basin brine, Lisbon Valley, Utah: fluid inclusion and isotopic evidence” Economic Geology, Vol. 81, pp. 1853-1866, 1986.
[18] I. R. Garden, I. R., S. C. Guscott, K. A. Foxford, S. D. Burley, J. J. Walsh, J. Watterson, “An exhumed fill and spill hydrocarbon fairway in the Entrada sandstone of the Moab anticline, Utah” in Migration and interaction in sedimentary basins and orogenic belts: Second International Conference on Fluid evolution, J. Hendry, P. Carey, J. Parnell, A. Ruffell, and R. Worden, eds. Belfast, Northern Ireland, p. 287–290.
[19] L.E. Spangler, D. L. Naftz, and Z. E. Peterman, “Hydrology, chemical quality, and characterization of salinity in the Navajo aquifer in and near the Greater Aneth oil field, San Juan County, Utah” USGS Water-Resources Investigations Report 96-4155, pp.96.
[20] D.B. Loope, R.M. Kettler, K.A. Weber., ‘Follow the water: Connecting a CO2 reservoir and bleached sandstone to iron-rick concretions in the Navajo Sandstone of south-central Utah, USA.” Geology, Vol: 38, No: 11, pp. 999-102, Nov 2010.
[21] G.F. Moulton, “Some features of redbed bleaching” AAPG Bulletin, Vol. 10, pp.304-311, 1922.
[22] D.W. Levandowski, M.E. Kaley, S.R. Silverman, R.G. Smalley., “Cementation in Lyons sandstone and its role in oil accumulation, Denver Basin Colorado” AAPG Bulletin, Vol. 57, pp.2217-2244, 1973.
[23] D.B. Segal, M.D. Ruth, L.S. Merin., “Remote detection of anomalous mineralogy associated with hydrocarbon production, Lisbon Valley, Utah” The Mountain Geologist, Vol. 23, pp. 51-62, 1986.
[24] R.C. Surdam, Z.S. Jiao, D.B. MacGowan, “Redox reactions involving hydrocarbons and mineral oxidants: A mechanism for significant porosity enhancement in sandstones” AAPG Bulletin, Vol. 77, pp. 277-286, 1993.
[25] Z.R. Beydoun, H.V. Dunnington. “The petroleum geology and resources of the Middle East: A Review” Journal of Petroleum Geology, Vol. 9, No. 1, Jan 1986.
[26] J.E. Huntoon, P.L Hansley, N.D. Naeser., “The search for a source rock for the giant tar sand triangle accumulation south eastern Utah” AAPG Bulletin, Vol. 83, pp. 467-495, 1999. BIETLER 2005
[27] V.F. Nuccio, S.M. Condom “Burial and thermal history of the paradox basin, Utah and Colorado and petroleum potential of the middle Pennsylvanian Paradox Basin” USGS Bulletin 2000-O, pp. 41, 2000.
[28] W.H. Craddock, M.S. Blondes, C.A. DeVera, A.G. Hunt., “Mantle and crustal gases of the Colorado Plateau: Geochemistry, sources and migration pathways” GCA, Vol. 213, pp 346-374, Sep 2017.
[29] Z.K. Shipton, J.P. Evans, B. Dockrill, J. Heath, A. Williams, D. Kirchner, P.T. Kolesar., “Natural leaking CO2-charged systems as analogues for failed geologic storage reservoirs” in carbon dioxide capture for storage in deep geologic formations, Eds. 2, D.C. Thomas and S.M. Benson. Amsterdam, Elsevier, pp-699-712.
[30] R.S. Haszeldine, O. Quinn, G. England, M. Wilkinson, Z.K. Shipton, J.P. Evans, J. Heath, L. Crossey, C.J. Ballentine, C.M. Graham., “Natural geochemical analogues for carbon dioxide storage in deep geological porous reservoirs, a United Kingdom perspective.” Oil & gas Science and Technology, Vol. 60, pp. 33-49, 2005.
[31] G. Purser, R.J. Rushton, J.M. Pearce, D. Wagner., “An Experimental and analogue study of iron release from red sandstones” Energy Procedia, Vol. 63, pp. 3268-3274, 2014.
[32] M. Wigley, N. Kampman, B. Dubacq, M. Bickle., Fluid-mineral reactions and trace metal mobilization in an exhumed natural CO2 reservoir, Green River, Utah” Geology, Vol. 40, No. 6, pp. 555-558, 2012.

Understanding the Paleofluid Records: a Case Study of Southern Utah by Rebecca Tyne is licensed under a Creative Commons Attribution 4.0 International License.
<< Back to Contents
<< Back to Publications
St Anne's Academic Review (STAAR) A Publication by St Anne's College Middle Common Room ISSN 2048-2566 (Online) ISSN 2515-6527 (Print)


Leave a Reply
You must be logged in to post a comment.